Below is the summary in French of the English report, prepared by the Environmental Chemistry Laboratory of Aix Marseille University and CNRS: “The purification performance of LaVie system: effect of free chlorine and dissolved oxygen concentration“
The purification performance of the LaVie system: effect of free chlorine and dissolved oxygen concentration

Notes: We have deliberately placed the conclusion under the introduction to allow for a quick understanding of the results of this study without going through the more technical part.
Introduction and Objectives
The LAVIE system aims to provide pure and high-quality water, both pleasant to drink and beneficial for health, while offering a drinking water solution for developing countries. This system purifies network water containing chlorine while eliminating bacteria and viruses. The report focuses on demonstrating the effectiveness of the LAVIE system in degrading organic compounds through photolysis (i.e., by direct light absorption) and explores the effectiveness of *free chlorine in this process.
Conclusion
The LAVIE system clearly demonstrates its effectiveness in degrading organic compounds in the presence of free chlorine and oxygen. The photolysis of chlorine and the production of reactive oxygen species play a crucial role in this process, making the LAVIE system an innovative method for purifying tap water by eliminating traces of pharmaceutical compounds and other organic pollutants.
Study Summary
Materials and Analytical Methods
- Products and Reagents: Use of *Diclofenac (DCF) as a reference compound, bleach, and other specific chemicals for analyses.
- Solution Preparation: DCF solutions prepared with ultra-pure water and mixed with tap water to obtain different concentrations of DCF and free chlorine.
- LaVie Purification System: Equipped with UV-A LEDs for a programmed exposure time of 15 minutes.
Analytical Methods
- Measurement of free chlorine and dissolved oxygen: Use of photometer and specific electrodes for these measurements.
- Reduction of organic compounds: Monitored by high-performance liquid chromatography (HPLC).
Results
- Experimental Parameters: Adjustment of DCF and free chlorine concentrations to observe significant degradation of organic compounds.
- Effect of free chlorine concentration on DCF degradation:
- High DCF concentrations: Enhanced degradation of DCF in the presence of free chlorine, with a linear relationship observed between chlorine concentration and percentage removal of DCF.
- Low DCF concentrations: Improved removal of DCF with increasing free chlorine concentration, confirming the involvement of chlorine in the degradation process.
- Effect of oxygen concentration: Experiments conducted under aerobic and anaerobic conditions show that the removal of oxygen significantly decreases DCF degradation, suggesting the involvement of reactive oxygen species in the process.
References
One of the figures illustrating the study
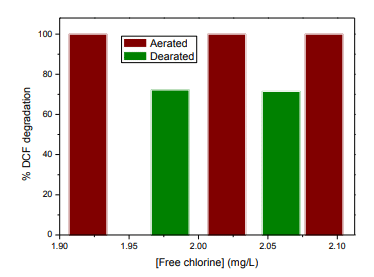
Caption: Degradation of DCF as a function of free chlorine concentration in tap water enriched with bleach after irradiation in the LAVIE system under *aerobic (Aerated) and anaerobic (Dearated) conditions, [DCF] = 2.5 mg/L and [free chlorine] approximately 2 mg/L.
Questions / Answers
*What is free chlorine?
Free chlorine refers to the chlorine available in water in the form of active chlorine, capable of disinfecting and destroying microorganisms. It primarily exists in two forms:
- Hypochlorous acid (HOCl): This is the most effective form for disinfection as it easily penetrates the cell walls of microorganisms.
- Hypochlorite ion (OCl⁻): This is a less effective form of free chlorine due to its ionic charge, which limits its ability to penetrate cells.
Free chlorine is often used in drinking water treatment to kill bacteria, viruses, and other pathogens, as well as to oxidize chemical contaminants. Its concentration is measured to ensure the effectiveness of disinfection while minimizing the formation of undesirable by-products such as trihalomethanes (THM).
What is the relationship between bleach and free chlorine?
Bleach and free chlorine are related but are not exactly the same thing.
Bleach:
- Bleach is an aqueous solution containing sodium hypochlorite (NaClO). It is a chemical product primarily used as a disinfectant and bleaching agent.
- When dissolved in water, sodium hypochlorite dissociates into hypochlorite ions (OCl⁻) and hypochlorous acid (HOCl), which contributes to its disinfecting action.
Free chlorine:
- Free chlorine in water refers to the active forms of chlorine, mainly hypochlorous acid (HOCl) and hypochlorite ion (OCl⁻).
- It is the free chlorine that is responsible for the disinfecting effect in treated water. The term “free chlorine” is used to measure the amount of chlorine available for disinfection in drinking water or pool water.
Relationship between the two:
- When bleach is added to water, it releases free chlorine in the form of hypochlorous acid (HOCl) and hypochlorite ions (OCl⁻).
- In this sense, bleach is a source of free chlorine when added to water.
Summary:
– Bleach = source of free chlorine (sodium hypochlorite in solution).
– Free chlorine = active forms (HOCl and OCl⁻) of chlorine in water, derived from bleach or other sources of chlorine.
Why was diclofenac (DCF), considered toxic, included in this experiment?
- Toxicity: Diclofenac, a commonly used non-steroidal anti-inflammatory drug (NSAID) for treating pain and inflammation, is often detected in surface waters and wastewater due to its widespread use and excretion by the body. Even at low concentrations, it can have toxic effects on aquatic life, particularly on fish and aquatic invertebrates.
- Presence in tap water: Although generally at very low concentrations, diclofenac can be found in tap water, resulting from the inability of wastewater treatment plants to completely remove pharmaceutical residues.
- Inclusion in the experiment: Diclofenac was used in this study as a reference compound to evaluate the effectiveness of the LAVIE system in degrading organic contaminants present in tap water. This demonstrates the system’s ability to remove traces of pharmaceutical products, thereby improving the quality of drinking water.
Why were the experiments conducted under aerobic (aerated) and anaerobic (dearated) conditions?
Experiments were conducted under aerobic and anaerobic conditions to assess the impact of dissolved oxygen on the effectiveness of the LAVIE purification system.
- Aerobic (aerated): The water contains dissolved oxygen, promoting the formation of reactive oxygen species (ROS) such as hydroxyl radicals, which effectively degrade contaminants.
- Anaerobic (dearated): The water is devoid of dissolved oxygen, which limits the formation of ROS and reduces the effectiveness of contaminant degradation.
Conclusion: The presence of dissolved oxygen significantly enhances the degradation of diclofenac by the LaVie system, demonstrating the importance of aerobic conditions (i.e., with the normal oxygen level found in tap water) for optimal purification.